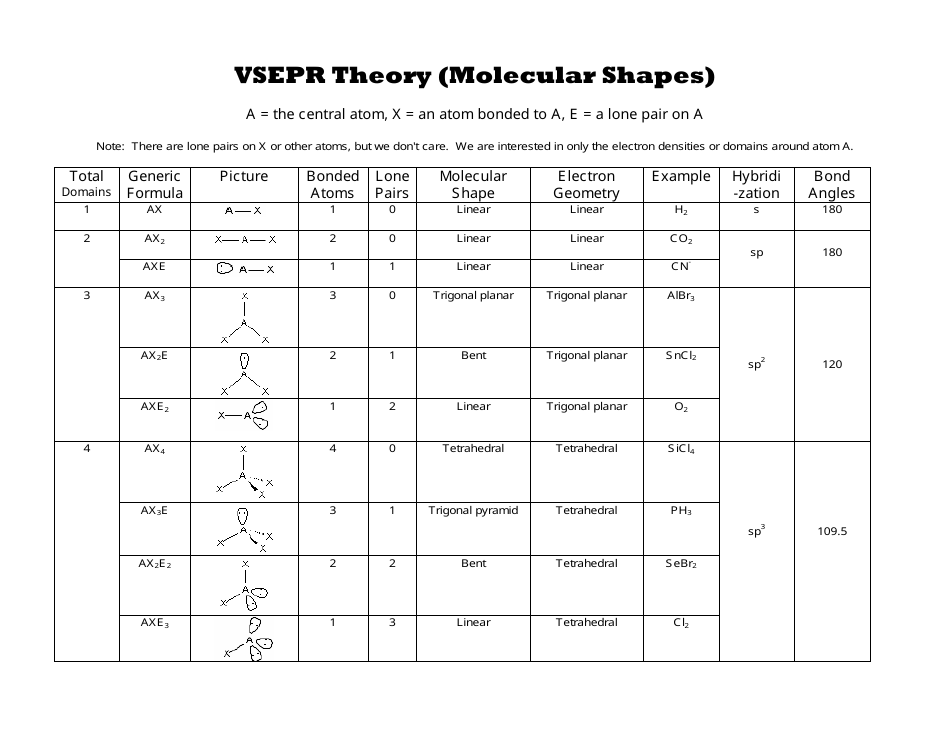
Fluorine belongs to the family of halogen in group 17 and has a valency of 7. Oxygen belongs to group 16, the chalcogen family, and has a valency of 6. The first and foremost step is to calculate the total number of valence electrons in an OF2 molecule. Step 1: Find the Total number of Valence Electrons. Flex Point Education Cloud™ is a trademark of Florida Virtual School. Steps to form OF2 Lewis Structure Diagram. | H 4 Lone Pairs: 0 Electron Geometry: Tetrahedral Br 2 Br – Br Total Domains: 1 Bonded Atoms: 1 Lone Pairs: 0 Molecular Shape: Linear Electron Geometry: Linear 180 ̊ Br Br PCl 3 C P C C Total Domains: 4 Bonded Atoms: 3 Lone Pairs: 1 Molecular Shape: Trigonal Pyramid Electron Geometry: Tetrahedral P C C C CH 3 Br H H C B H Total Domains: 4 Bonded Atoms: 4 Lone Pairs: 0 Molecular Shape: Tetrahedral Electron Geometry: Tetrahedral H H C B H Your Own Molecule CO 2 O = C = O Total Domains: 2 Bonded Atoms: 2 Lone Pairs: Molecular Shape: Linear Electron Geometry: O = C = O Unless Otherwise Noted All Content © 2022 Florida Virtual School. Flex Point Education Cloud™ is a trademark of Florida Virtual School. Before we get to the geometry of molecules with lone pair(s), you need to know that a structure is characterized by molecular and electron geometry, which sometimes may be the same. Compoun d Draw Lewis Structure Use the VSEPR Chart to determine: Determine the structures of the molecule Draw the VSEPR model H 2 O Total Domains: 4 Bonded Atoms: 2 Lone Pairs: 2 Molecular Shape: Bent Electron Geometry: Tetrahedral SiH 4 H | H-Si-H Total Domains: 4 Bonded Atoms: Molecular Shape: Tetrahedral H H Si H H Unless Otherwise Noted All Content © 2022 Florida Virtual School. Let’s put a little chart of example for steric number ranging from 2-6. Activity: The first row is done for you as a model. Intermolecular forces also affect molecular shape. Covalent bonds impose constraints on atom arrangement, while ionic bonds create crystal structures. Electron molecular geometry: chemistryMolecular and electronic geometry chart. How does the force between the atoms impact the shape of a molecule? The shape of a molecule is influenced by the force between atoms, which is determined by the type of chemical bonds. The VSEPR theory predicts a molecule' s shape based on electron pairs around the central atom, considering the number of electron pairs and the presence of lone pairs. Electron domain geometry definitionMolecular geometry and electron geometry chart Geometry molecular electronic chart electron bonding bond groups anglesGeometry molecular chart chemistry shapes molecules vsepr general pdf organic worksheet sampletemplates worksheets polarity explore bonding. Review Questions Write a statement explaining which characteristics of an atom determine the VSEPR shape of a molecule (hint: look at the VSEPR geometry chart). The ideal bond angles in a trigonal pyramid are based on the tetrahedral electron pair geometry.Molecular Structure Instructions: Fill in each section of this worksheet and submit it to your instructor for grading. \)) with the nitrogen atom at the apex and the three hydrogen atoms forming the base. Study with Quizlet and memorize flashcards containing terms like electronic geometry: linear molecular geometry: linear, electronic geometry: trigonal planar molecular geometry: trigonal planar, electronic geometry: trigonal planar.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
